Summary: The Supreme Court of India, led by Judge Ahsanuddin Amanullah, made a decision on a case about incorrect labels on vaccines made by Panacea Biotec Ltd. The case was about mistakes in vaccine labels, which led to accusations of false labeling under the Drugs and Cosmetics Act, 1940.
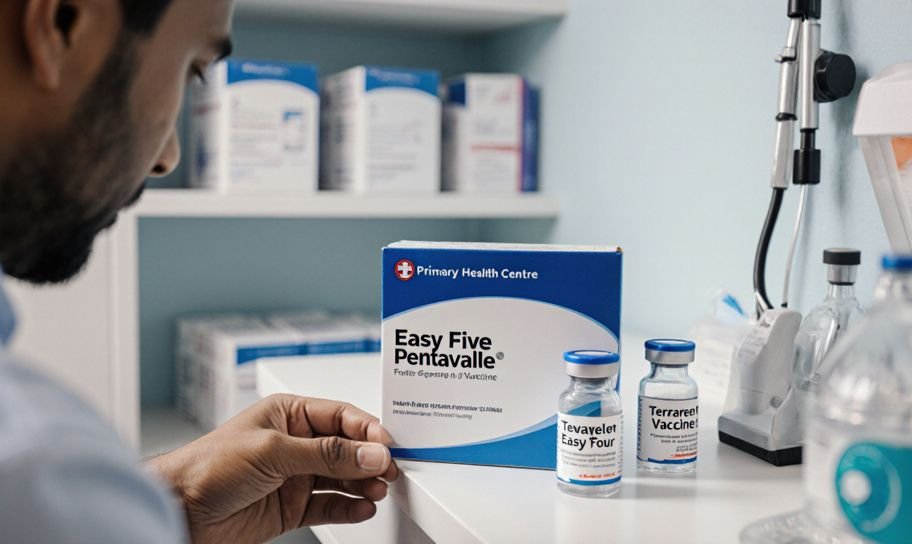
On October 21, 2005, Mr. Joy Mandi found a labeling problem with a vaccine from Panacea Biotec Ltd. He reported this to the Primary Health Centre in Thrissur, Kerala. The box said the vaccine was "Easy Five Pentavalent," but the bottle inside was labeled "Tetravalent Vaccine Easy Four."
The complaint pointed out that the box said the vaccine had five ingredients, including a Hepatitis vaccine, but the bottle inside said it only had four ingredients, leaving out the Hepatitis vaccine. This mistake led to accusations of false labeling.
"According to the label on the box, the drug is Easy Five pentavalent vaccine... But according to the label on the bottle Easy Four tetravalent vaccine... does not contain HbSAg 10 mcg." - Complaint Document
Judge Ahsanuddin Amanullah, along with Judge R. Mahadevan, reversed the High Court's decision, stressing that the steps were followed correctly because the complaint was made by a government worker. The Supreme Court brought back the complaint, allowing the trial to continue.
The Supreme Court decided that the trial against Panacea Biotec Ltd. should continue, emphasizing that the complaint was valid because it was filed by a public servant. This ruling highlights the need for accurate vaccine labeling and the importance of following legal procedures in such cases.